“This is very good news. We know that in the next few months Norway will reach a peak in the number of patients who are ill with COVID-19. With these emergency ventilators the health service will be better equipped to handle such a peak,” said Prime Minister Erna Solberg.
GOVERNMENT PROVIDING FUNDS
She said she is impressed by the companies that have leaped into action to meet an urgent challenge.
“This shows what we can achieve in this country when it really counts. Different environments have come together and used their expertise in new ways. We welcome all initiatives and creative suggestions to ensure that everyone receives high-quality treatment. We must keep two tasks in mind at all times: doing what we can to fight the virus, and working to increase health service capacity in case many people become ill at the same time,” said Solberg.
The Government is therefore guaranteeing project funding and has ordered 1,000 emergency respirators. The production of emergency ventilators in Norway is a collaboration between the Defence Research Establishment, Servi and Laerdal Medical.
MAY BE EXPORTED
The Norwegian Institute of Public Health has estimated a possible need for between 600 and 1 200 intensive care units in Norway. Norway currently has 682 respirators, and more will be delivered in the coming weeks.
‘If the project goes according to plan, production could start as early as April. Most patients in intensive care will need breathing assistance,’ said Bent Høie, Minister of Health and Care Services.
The COVID-19 epidemic has triggered a critical need for respirators worldwide, so delivery times estimated by the manufacturers are longer and more uncertain than normal.
“We have ordered more emergency ventilators from Laerdal Medical than we need in Norway. We know other countries also have a great need and are therefore looking at the possibility of export,” said Høie.
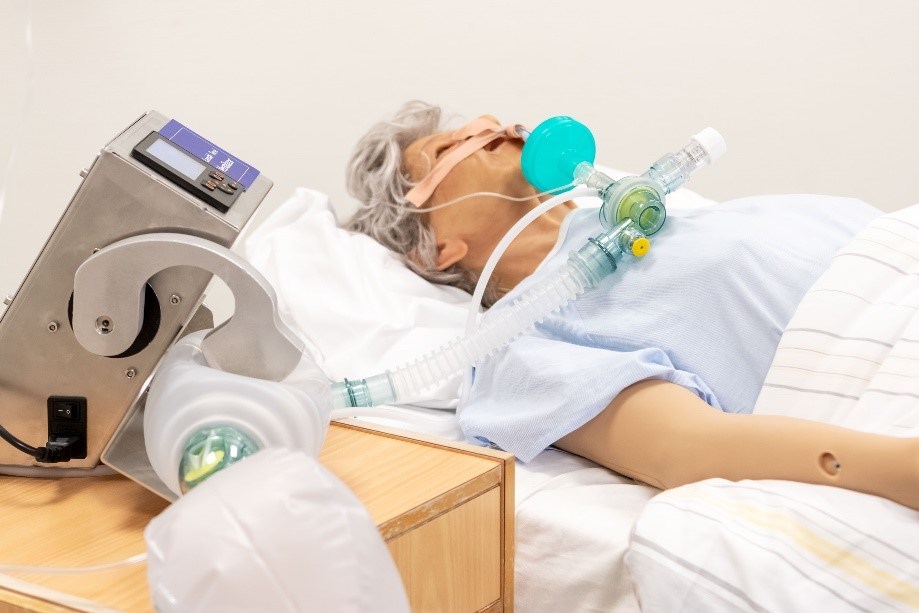
ABOUT EMERGENCY VENTILATORS
In record time, the Defence Research Establishment, Laerdal Medical and Servi have developed a new emergency ventilator for intensive-care patient treatment. The product improves lung functioning by means of a breathing balloon with valves and alarms to ensure against excessive pressure. The product has been evaluated and recommended by clinical experts at the university hospitals in Stavanger and Oslo.